NUQUATIC ADVANTAGE™
PFAS Technology
NuQuatic’s ADVANTAGE™ process is designed to address one of the most critical challenges in PFAS treatment: How to efficiently remove PFAS from water while minimizing the cost and complexity of final destruction. By integrating concentration and destruction into a single treatment approach, NuQuatic enables a more complete and cost-effective PFAS management solution.
How The Process Works
PFAS are persistent synthetic chemicals found as contaminants in water sources around the world. NuQuatic’s patented technology concentrates and destroys PFAS through a four-step process.
Using the unique properties of aluminum, the process enhances PFAS capture and concentration through foam fractionation, resulting in very high PFAS concentration factors using non-toxic, recyclable, and cost-efficient materials. The concentrated PFAS stream is then treated in an integrated electrolytic cell uniquely optimized for destruction.
As shown in the process diagram, the system consists of four key steps:
-
Mixing: Aluminum is introduced and mixed with incoming water
-
Primary Concentration: PFAS are captured through foam fractionation
-
Secondary Concentration: Additional concentration under optimized conditions
-
Destruction: Electrolytic treatment of the concentrated PFAS stream
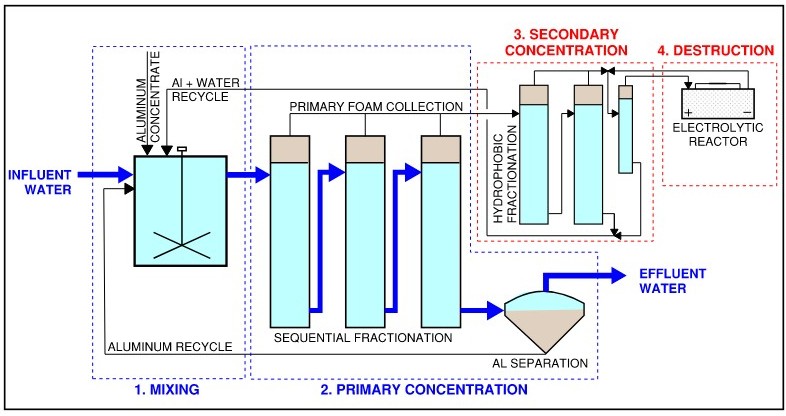
The NuQuatic ADVANTAGE™ Process for Removal and Destruction of PFAS in Water
KEY ADVANTAGES
Applications
NuQuatic technology is designed for a wide range of PFAS-impacted water sources:
- Municipal drinking water and wastewater
- Industrial wastewater
- Energy production and power generation sites
- AFFF-contaminated groundwater
- Lanfill leachate and high-strength PFAS streams
Performance
- Demonstrated high PFAS concentration factors (up to ~100,000x)
- Proven PFAS removal efficiency below MCL requirements for regulated PFAS
- Greater than 99% destruction of regulated PFAS and more than 86% destruction of all detected PFAS compounds
(Note: performance dependent on site-specific conditions)

Facilities & Capabilities
At NuQuatic’s Clawson, Michigan facility, we support technology development and demonstration across multiple scales. Our reconfigurable test platforms operate at gallon-per-day scale flow rates and are supported by wet lab and analytical capabilities, including high-precision LC-MS/MS for rapid PFAS analysis.
For field applications, NuQuatic deploys a mobile demonstration trailer capable of gallon-per-minute scale treatment, enabling on-site pilot testing and performance validation under real-world conditions.
TECHNOLOGY + PERFORMANCE COMPARISON
See how our technology and performance compares to industry options.
| Removal & Complete Destruction | (3) | (3) | (3) | (3) | (3) | (3)(5) | (3) |
|
| Low Cost Solution | (1) | (2) | ||||||
| Low PPT Effluent | ||||||||
| Compact Footprint | ||||||||
| Scalable: Leachate to Potable | (5) | |||||||
| Rapid Deployment | ||||||||
| Minimal Waste Generation | (4) |
(1) Including Cost of Carbon Disposal
(2) Including Cost of IX Resin Disposal
(3) No Destruction
(4) No PFA Waste
(5) No PFA Removal
Meets Criteria
Meets Some
Does Not Meet
(1) Including Cost of Carbon Disposal
(2) Including Cost of IX Resin Disposal
(3) No Destruction
(4) No PFA Waste
(5) No PFA Removal
Meets Criteria
Meets Some
Does Not Meet



